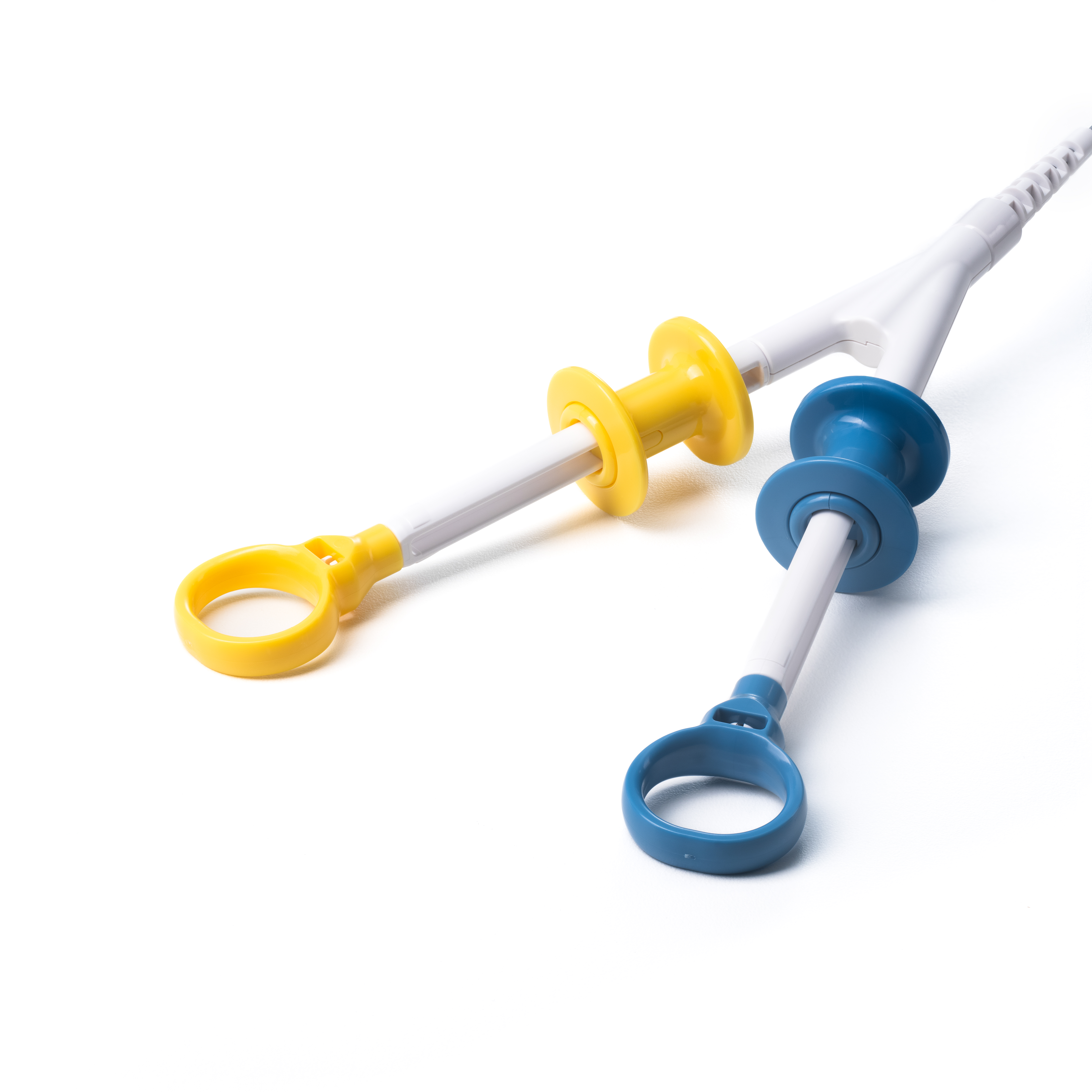
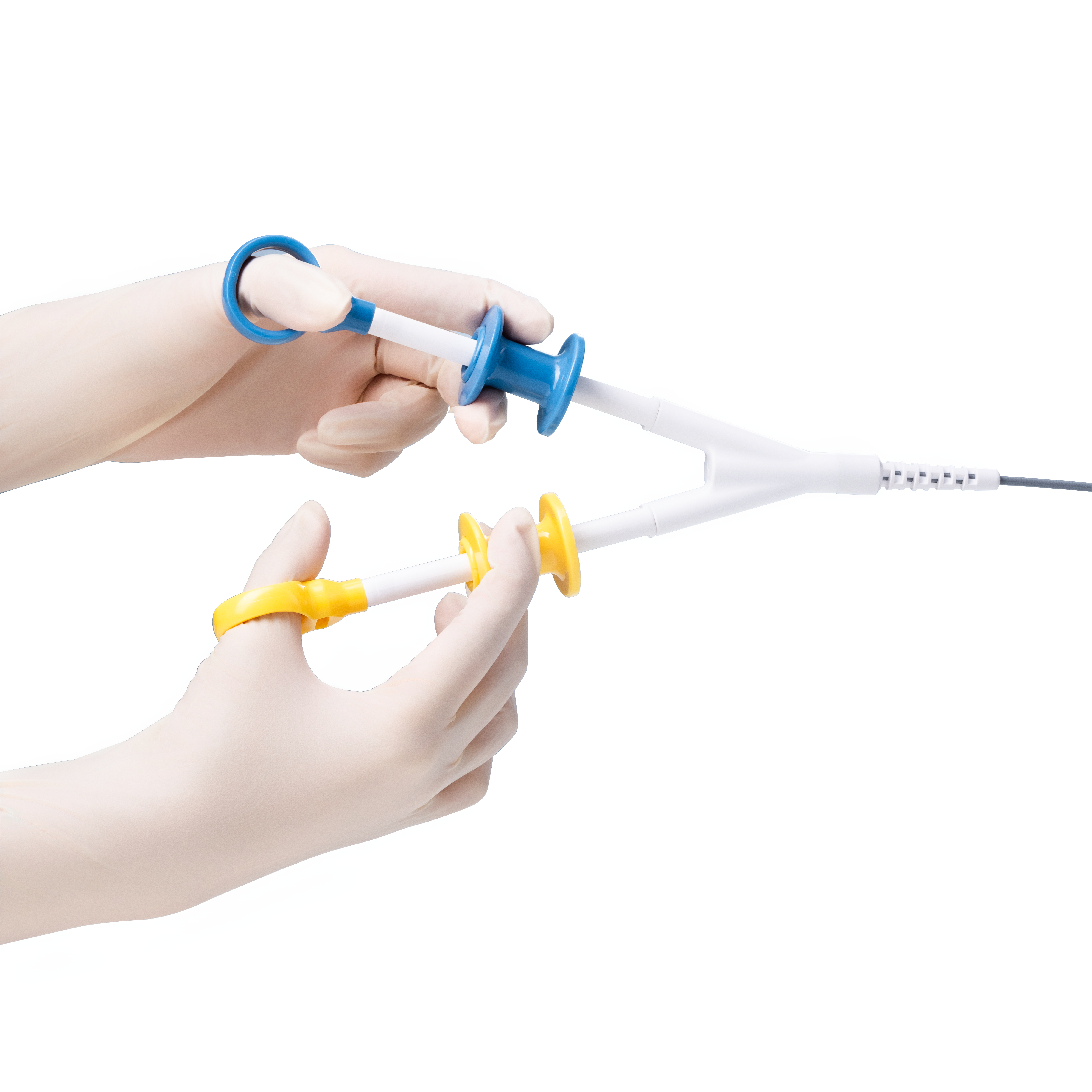
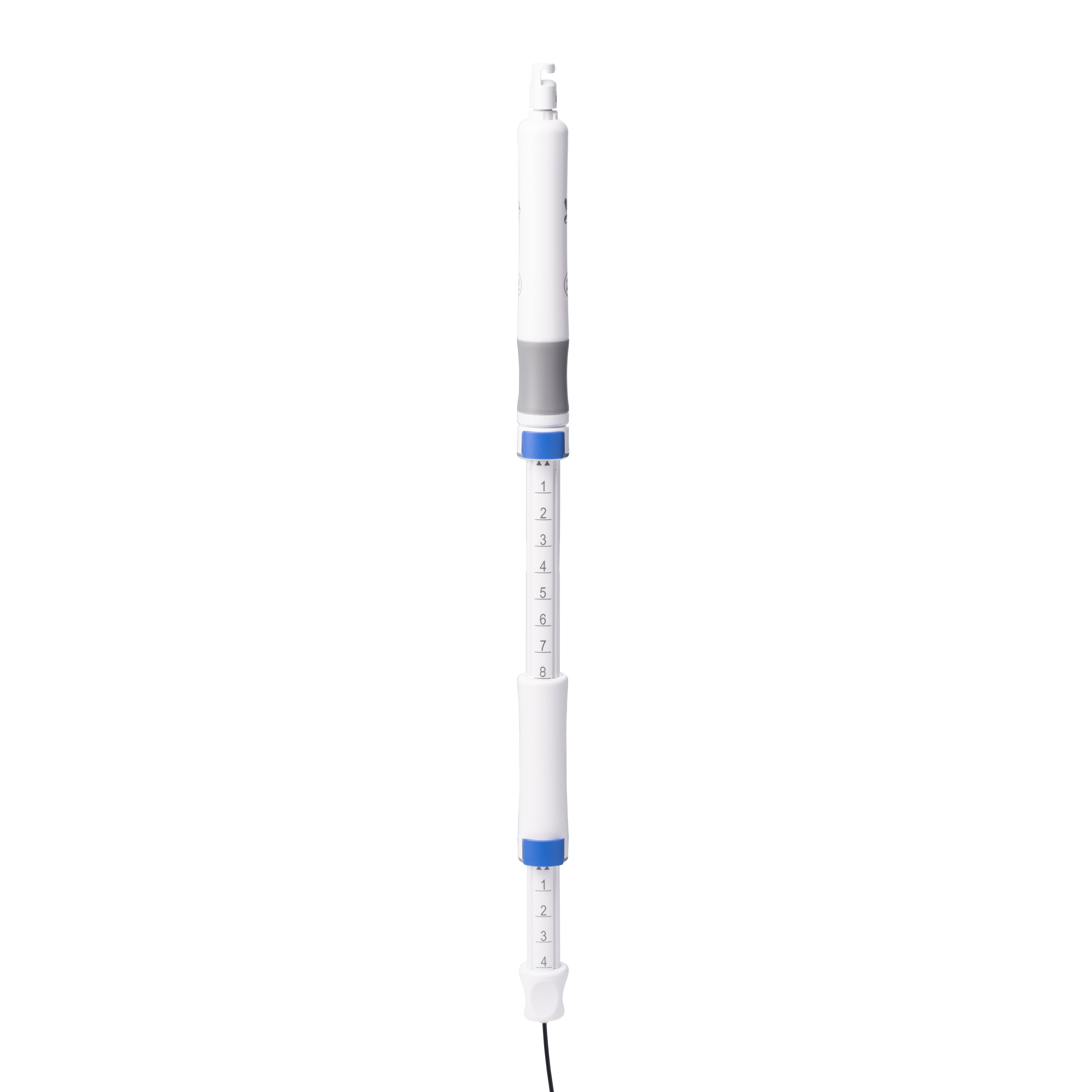
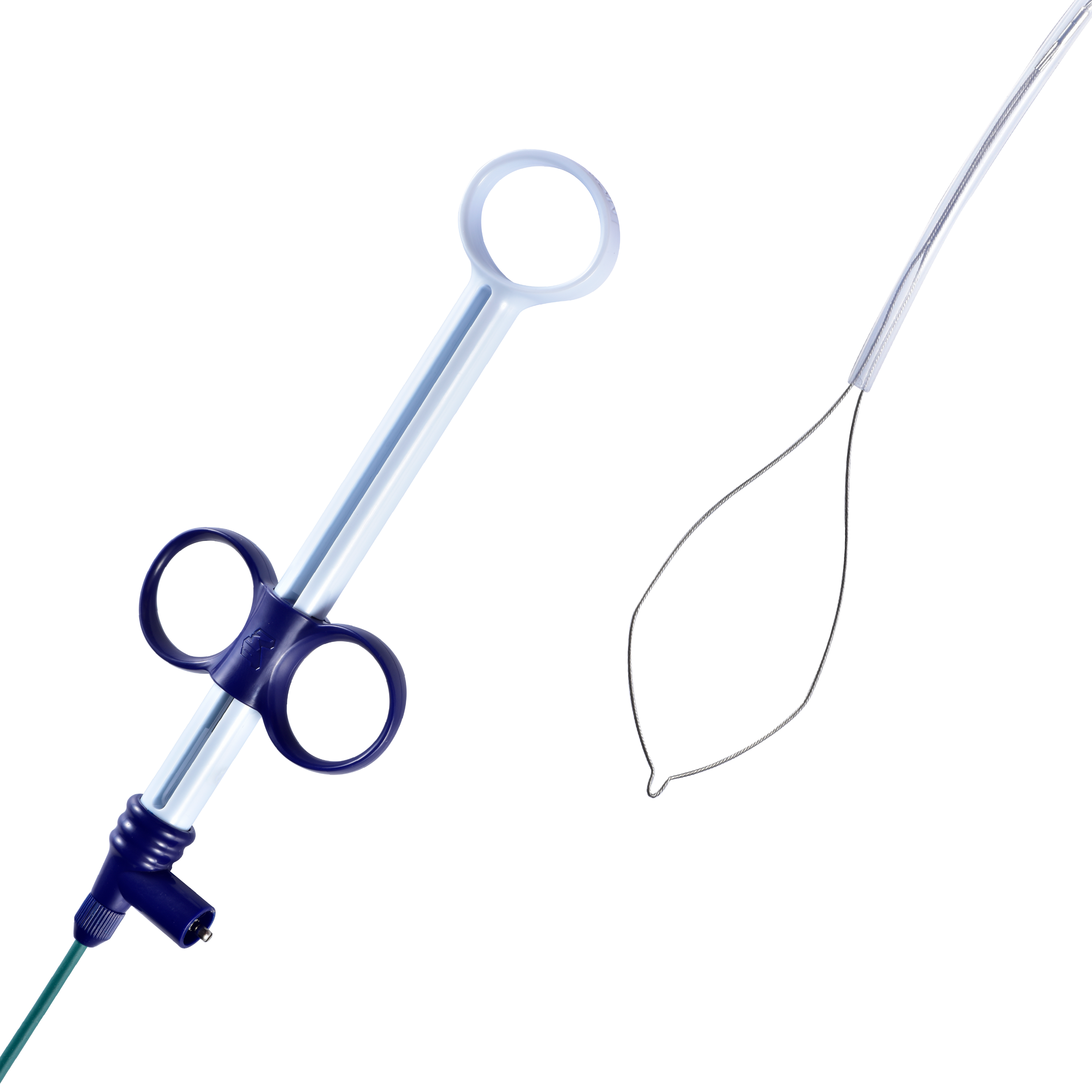
Sebastian Antonius Brocke is Vice President of Finance for MICRO-TECH in EMEA, responsible for Finance, IT, HR, Administration, Supply Chain, and Customer Service across MICRO-TECH Europe GmbH and the EMEA Regional Headquarters. With over a decade of experience spanning production, service, and sales organizations, he plays a key role in strengthening the company’s financial strategy, operational efficiency, and regional governance.
Sebastian brings deep expertise in enterprise architecture, process optimization, IT governance, and digital transformation. Combining analytical rigor with hands-on pragmatism, he bridges strategic vision with operational execution to deliver sustainable results.
Throughout his career, Sebastian has led cross-functional teams, overseen multimillion-euro transformation programs, and delivered measurable improvements in efficiency, agility, and value creation. Known for his structured and collaborative leadership style, he is a trusted partner in building agile, future-ready organizations across the EMEA region.